Description
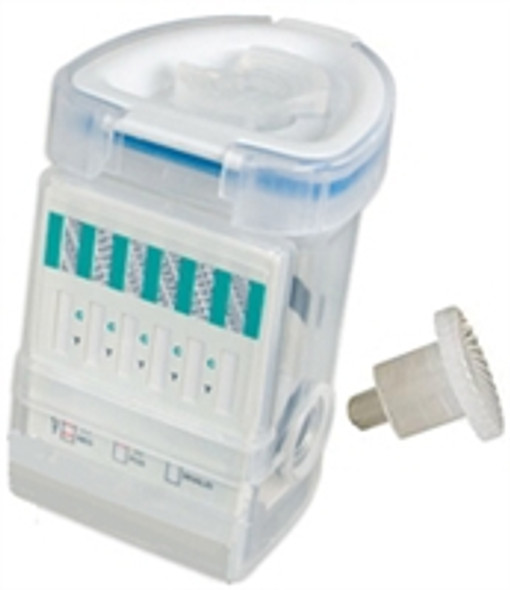
6 Panel E-Z Split Key Specimen Urine Drug Test Cup
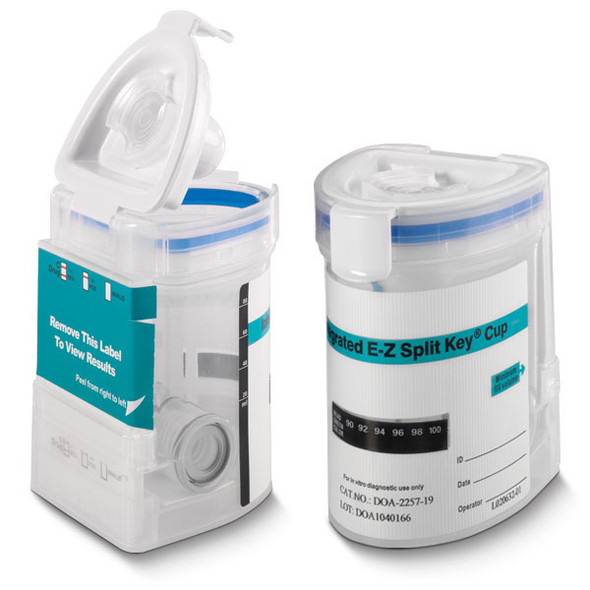
This FDA Moderately Cleared Cup is Collector Controlled not Donor Controlled, meaning that the collector is the only person who has complete control of the device and can activate the test at their convenience. Once the test is performed and the results are displayed, the collector can make a photocopy of the result simply by placing the flat front side onto a copy machine or scanner. The E-Z Integrated Split Key Cup from Abbott Diagnostics formerly Alere Toxicology / Instant Technologies is the first rapid response onsite testing device to offer a compact, split specimen format that is not only easy to use, but is easy to administer because of its compact and oval shape front and squared back, the E-Z Key Cup is easy to use for men and women. Split Specimen E-Z Cup is an innovative drug test cup design that eliminates urine handling and donor tampering, providing a unique and efficient approach for on-site testing. The E-Z Integrated Split Specimen drug test is initiated when the key is inserted into the cup which allows the operator to run drug tests at your convenience.
The Integrated Split Key Cup is the first immediate response onsite testing device to offer a compact, split-specimen, fully integrated, self contained drug screening cup for detecting multiple drugs and drug metabolites existence in urine with the accuracy of laboratory tests in a shorter time. The E-Z integrated split specimen Key cup can be ordered with the combinations from 3 to 12 panel test cups. The test utilizes monoclonal antibodies to selectively detect elevated levels of specific drugs in urine. The Integrated E-Z Split Key Cup is a lateral flow chromatographic immunoassay for the qualitative detection of multiple drugs and drug metabolites in urine.
Features
- Secure "Key" activation
- Fast Results
- Reserved Split Specimen Ideal for When Additional Confirmation Testing is Needed
- Adheres to SAMHSA Cutoff Levels
- Cup Contains Temperature Strip to Ensure Specimen Validity
- Easy-To-Use & Quality Urine Drug Test Cup
- Flat Sided Cup for Ease of Photocopying Results
- Up to 99% Accurate Results in 5 minutes
- The E-Z Key Cup specimen collector can activate the test with special key eliminating donor manipulation
- The E-Z Key Cup Label hides test result from those who do not need to view results
- Fully Integrated cup design eliminates handling of the urine
- 12 to 18 Month Shelf Life
- FDA Cleared for Professional Use
- Available Adulteration Detection configurations
- No handling, dipping, tilting, or touching of the urine is required
- Leak proof snap top lid Integrated Temperature Strip Auto-Split sample for specimen integrity
- The Split Specimen cup detects multiple drugs existence with laboratory accuracy
- 25 Cups Per Box
| 6 Panel EZ Split Specimen Key Cup | ||
| Item Number | Panel Configuration | Adulterants |
| DOA-1167-019 | AMP, COC, mAMP, OI, PCP, THC | NONE |
| DOA-2167-019 | AMP, BZO, COC, mAMP, OPI, THC | NONE |
| DOA-167-321-019 | COC, mAMP, MDMA, OPI, OXY, THC | NONE |
| E-Z Split Key Cup Detection Times & Cut-off Levels in Urine | |||||
| Drug Name | Abbreviation | Calibrator | Cutoff | Minimum Detection |
Maximum Detection |
|---|---|---|---|---|---|
| Amphetamine | AMP | d-Amphetamine | 1000 ng/ml | 2-7 hours | 2-4 days |
| Barbiturates | BAR | Secobarbital | 300 ng/ml | 2-4 hours | 1-3 weeks |
| Benzodiazepines | BZO | Oxazepam | 300 ng/ml | 2-7 hours | 1-4 days |
| Buprenorphine | BUP | Buprenorphine | 10 ng/ml | 2-7 hours | 2-3 days |
| Cocaine | COC | Benzoylecgonine | 150/300 ng/ml | 1-4 hours | 2-4 days |
| Ecstasy | MDMA | d,l-Methylenedioxymethamphetamine | 500 ng/ml | 2-7 hours | 2-4 days |
| Marijuana | THC | 11-nor-Delta9-THC-9 COOH | 50 ng/ml | 2 hours | up to 40 days |
| Methadone | MTD | Methadone | 300 ng/ml | 3-8 hours | 1-3 days |
| Methamphetamine | mAMP | d-Methamphetamine | 1000 ng/ml | 2-7 hours | 2-4 days |
| Morphine | MOP (OPI-300) | Morphine | 300 ng/ml | 2 hours | 2-3 days |
| Opiates | OPI | Morphine | 2000 ng/ml | 2 hours | 2-3 days |
| Oxycodone | OXY | Oxycodone | 100 ng/ml | 1-3 hours | 1-2 days |
| Phencyclidine | PCP | Phencyclidine | 25 ng/ml | 4-6 hours | 7-14 days |
| Tricyclic Antidepressants | TCA | Nortriptyline | 1000 ng/ml | 8-12 hours | 2-7 days |
| Propoxyphene | PPX | Propoxyphene | 300 ng/ml | 2-7 hours | 2-3 days |
| * Detection times are not guaranteed. Please refer to panel configuration above for complete list of drugs tested for this device. This assay provides only a preliminary analytical test result. A more specific alternate chemical method must be used in order to obtain a confirmed analytical result. Gas Chromatography/Mass Spectrometry (GC/MS) is the preferred confirmatory method. Clinical consideration and professional judgment should be applied to any drug of abuse test result, particularly when preliminary positive results are indicated. | |||||
Reference Documentation - Download/Print
![]() E-Z Integrated Key Cup Drug Test Package Insert
E-Z Integrated Key Cup Drug Test Package Insert![]() E-Z Integrated Key Cup Drug Test Procedure Card
E-Z Integrated Key Cup Drug Test Procedure Card
E-Z Split Key Cup Instructions for Use
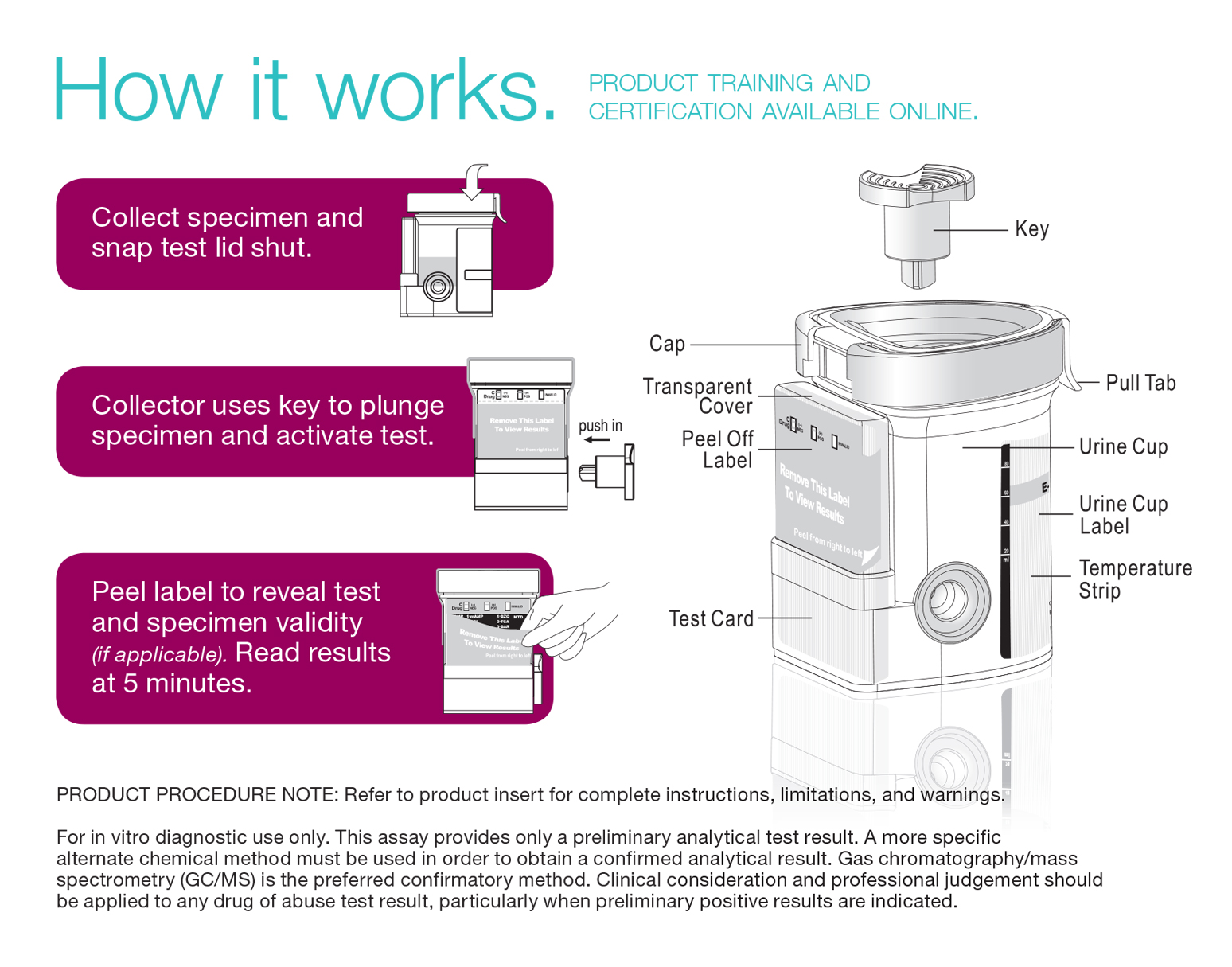
- Remove the key by twisting it from the center of the cup cap. Open cap with the Pull Tab.
- Have the donor urinate directly into the cup and secure teh cap, making sure to provide a urine sample that's a minimum of 30ml. Note: Secure cap by pressing down on the Pull Tab until an audible click is heard.
- Once the sample is returend to the collection official check the cap for a tight seal. Collection official dates and initials the security seal and places the security seal over cap.
- On a flat surface, collection official inserts key and pushes in. Peel back the label to reveal the test panels and read the results at 5 minutes. Note: If the test cup includes Adulteration tests read results between 3-5 minutes. Results read after 10 minutes should be considered invalid
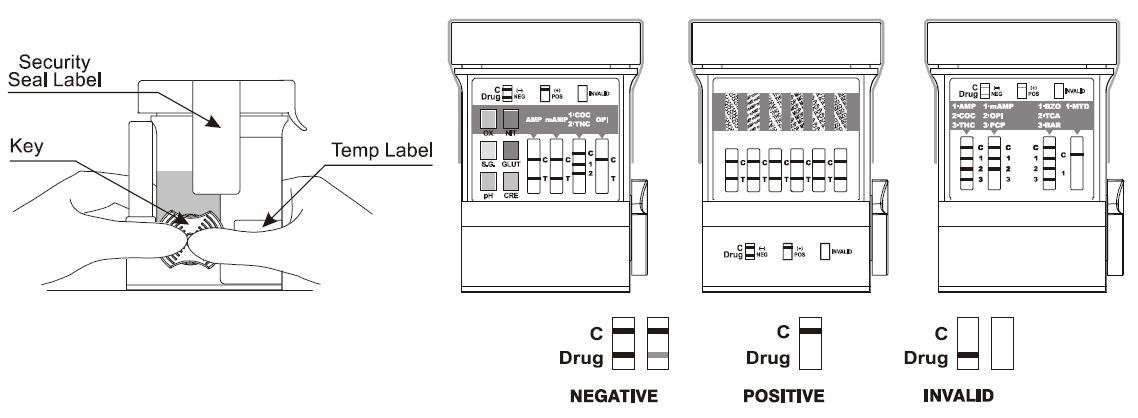
How to Read and Interpret Drug Test Results
- Negative drug test results will have two lines - one line in the control region and one line in the test region.
- Presumptive positive results will show only one line in the control region. If a test fails to show a line in the control region, this indicates an invalid test result.
- Invalid drug test results occur when no line appears in the control region. Should this occur, repeat the test with a new device. If results are still invalid, please stop using the test cups and contact a TestCountry representative.