Description
CSI Cup - CLIA Screen In-Vitro 14-Panel CLIA Waived Drug Screening Test Cup with 3 Adulterants
This High Quality CLIA Waived drug test cup is an easy, fast, qualitative, competitive binding immunoassay method for screening without the need of instrumentation.  The best selling CSI Cup (CLIA Screen In-Vitro) Multi-Panel Drug Test Cup is a No Step, fully integrated self contained multi-drug screening cup. No tipping or turning required. Read results in 5 minutes. The cup is FDA Cleared and CLIA Waived. This cup is a fast visual, competitive panel immunoassay that can be used for the simultaneous, qualitative detection of multiple drug metabolites at specific cutoff levels in human urine.
The best selling CSI Cup (CLIA Screen In-Vitro) Multi-Panel Drug Test Cup is a No Step, fully integrated self contained multi-drug screening cup. No tipping or turning required. Read results in 5 minutes. The cup is FDA Cleared and CLIA Waived. This cup is a fast visual, competitive panel immunoassay that can be used for the simultaneous, qualitative detection of multiple drug metabolites at specific cutoff levels in human urine.
Features & Benefits
- CLIA Waived
- FDA 510(K) Approved
- Sold in boxes of 25 tests
- New "Click Seal" cap - when you hear the “CLICK” the cup is sealed and the specimen is secured
- Up to 99% Accurate
- Dual scale Temperature Strip verifies if urine is "fresh" and not diluted
- Adheres to the SAMHSA Cutoff Levels
- Immediate Results
- Shelf-life up to 24 months
- New easy to open top for the confirmation lab
Download & Print Reference Materials
![]() CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Package Insert
CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Package Insert
![]() CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Instruction Sheet
CLIA Screen In Vitro Cup (CLIA Waived) Drug Test Instruction Sheet
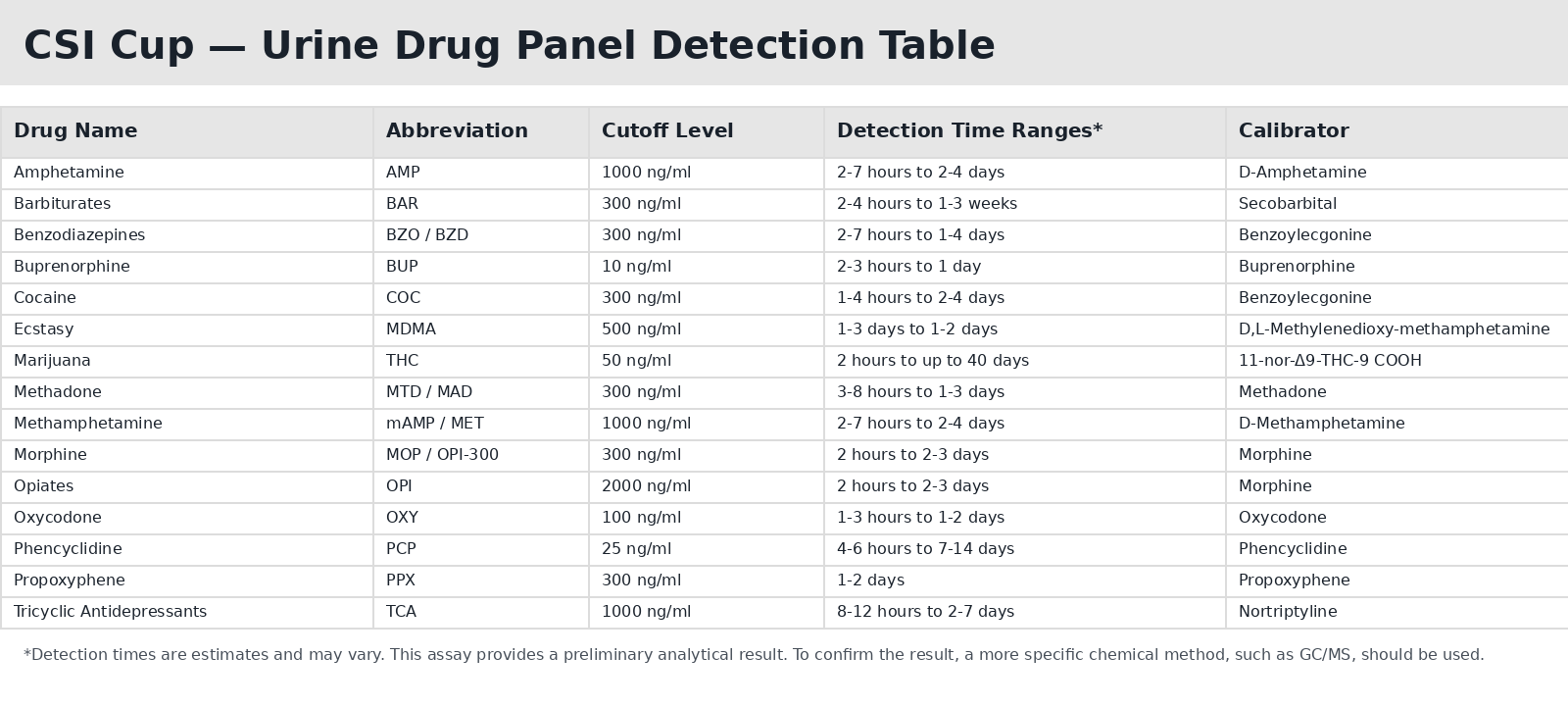
| Fourteen Panel Configuration | ||
| Item Number | Drugs Tested | Adulterants Tested |
| CSI-CUP-1145A3 | AMP, BAR, BUP, BZO, COC, MET, MDMA, MOP300, MTD, OXY, PCP, PPX, TCA, THC | OX - Oxidants SG - Specific Gravity PH - pH Level |